Drugs for Malaria: the Challenge of Resistance to Front Line Therapy in Control and Elimination
In honor of World Malaria Day, Kasturi Haldar, Editor-in-Chief of PLOS Pathogens comments on the challenge of resistance to frontline drugs in malaria treatment, control, and elimination on the path to eradication.
From 2010-2015 new malaria cases and malaria deaths in the world fell remarkably by ~20% and ~30% respectively. Yet a substantial global burden remains, with over 400,000 deaths and 200 million new cases reported in 2016. Presently drugs are the path to treat malaria infection and reduce the parasite burden and disease in patients. In particular, artemisinin combination therapies (ACTs) have played a central role against Plasmodium falciparum (the deadliest of human malaria parasites), but unfortunately, resistance to artemisinins has emerged and spread throughout Southeast Asia, casting a grim specter of losing on gains in malaria control and elimination.
The identification of markers and mechanisms of artemisinin resistance has greatly facilitated the development of new strategies to circumvent it. Parasites have emerged that are simultaneously, multi-drug resistant to both the artemisinins and the partner drug, suggesting need for new (non-artemisinin based but nonetheless) fast acting combinations. Further confounders to combating artemisinin resistance include a role for host factors and prevalence of resistance in asymptomatic (and therefore often undetected) infection in areas where parasite burdens are low but transmission still occurs. It is especially concerning that de novo emergence of resistance (rather than spread) has been reported in symptomatic presentation in an area of high endemicity in Africa. Drugs are needed to target Plasmodium vivax, a second, widespread parasite species with a latent liver stage infection that is not blocked by ACTs. Given that as malaria burdens decrease, antimalarial drugs have to eliminate malaria in the absence of blood stage immunity, we need to also reduce transmission by the mosquito vector, a critical focus of prevention strategies.
This is reflected in an ongoing PLOS Collection, Malaria: Targets and Drugs for All Stages, curated every two years (next update expected this summer) that highlights complexities of malaria disease, infection, and transmission in human infection, animal models, and the insect vector. The collection includes studies on global drug discovery efforts mechanisms of drug action and resistance. The papers cover a wide range of leading research, and although they present only a portion of the literature they reflect larger malaria drug discovery and mechanistic studies needed for new intervention strategies that address hurdles for malaria elimination.
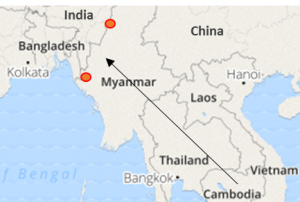
Since 2010, seven countries have been certified to have eliminated malaria (by achieving three consecutive years of zero locally-acquired malaria). They include United Arab Emirates (2007), Morocco (2010), Turkmenistan (2010), Armenia (2011), Maldives (2015), Sri Lanka (2016) and Kyrgyzstan (2016). Malaria elimination campaigns in India and Bangladesh are expected to be particularly important to stem the global spread of artemisinin and multi-drug resistant strains from Southeast Asia to the rest of the world (see Fig. 1). They may be empowered by fruits of a global, long-term drug discovery pipeline, that shows promise to yield new chemical agents (and their combinations) presently in advanced efficacy studies, expected to combat artemisinin resistance and deliver long lasting protection, possible in a single curative dose.
Kasturi Haldar, Ph.D. Editor-in-Chief, PLOS Pathogens, is the Julius Nieuwland Chair of Biological Sciences and Parsons-Quinn Director of the Boler-Parseghian Center for Rare and Neglected Diseases, University of Notre Dame, which supports private public partnerships and drug discovery in rare and neglected diseases, including malaria. She serves on the Expert Scientific Advisory Board of Medicines for Malaria Venture and review panels of the National Institutes of Health. She has received research funding from both organizations and the Bill and Melinda Gates Foundation.
Featured Image Credit: Frevert et al. (2005)
